What is Foam, and How Does It Affect Our Industry?
Have you ever wondered why foam shows up in your industrial processes, seemingly out of nowhere, causing headaches? It’s a common issue, and I’ve seen how this seemingly minor problem can silently disrupt operations.
Foam is a dispersion of gas bubbles in a liquid, forming a stable, persistent structure on the liquid's surface or throughout the bulk. It forms when gases become trapped within a liquid due to agitation, mixing, or chemical reactions1, often stabilized by surfactants2 or other surface-active substances present in the system.

I often explain to clients that foam is rarely just a surface nuisance. It's truly a process disruptor, and understanding it is the first step to controlling it.
What Is Foam in Manufacturing?
Do you know foam’s true nature in a manufacturing environment? It's more than just bubbles; it's a specific physical state that shows up in many industrial processes.
In manufacturing, foam is a colloidal system where gas is dispersed in a liquid, creating a transient or stable structure. Think of it like beating egg whites—you introduce air into the liquid protein, and it forms a stable, light structure. Similarly, in industrial settings, when air or other gases are introduced into a liquid, and that liquid contains certain stabilizing agents, foam can appear. These stabilizing agents are often called surfactants, but they can be other dissolved solids, polymers, or even fine particulates. The key is that these agents reduce the surface tension of the liquid and form a film around the gas bubbles, preventing them from coalescing and breaking quickly.

From chemical reactors to wastewater treatment plants, foam is a familiar, often unwelcome, sight.
How Is Foam Formed in Industrial Processes?
Have you ever wondered about the actual mechanics behind foam formation in your operations? It boils down to a few key factors that interact to trap gas.
Foam forms when three conditions combine in a liquid system: gas entrainment, a surface-active substance, and mechanical agitation3. First, gas must enter the liquid. This can happen through vigorous mixing, pumping, spraying, or even chemical reactions that produce gas. Imagine a high-speed mixer whipping air into a chemical solution. Second, the liquid must contain surface-active substances. These are molecules that prefer to sit at the interface between the liquid and gas, reducing the surface tension. They act like tiny shields around gas bubbles. Examples include surfactants, proteins, dissolved polymers, or even fine solid particles. Third, there must be a mechanism to generate and sustain the finely dispersed gas bubbles. This is typically mechanical agitation, turbulent flow, or pressure changes. When gas bubbles are formed and stabilized by these surface-active agents, they resist bursting and accumulate, leading to foam. The stability of the foam depends on the properties of the liquid, the gas, and the surface-active agents present.
| Factor | Description | Common Examples in Industry |
|---|---|---|
| Gas Entrainment | Introduction of gas (often air) into the liquid through physical means or chemical reactions. | High-speed mixing, vigorous pumping, cascading liquids, fermentation gas production. |
| Surface-Active Agents | Substances that reduce the surface tension of a liquid and stabilize gas-liquid interfaces, forming persistent films around bubbles. | Surfactants (detergents), proteins, naturally occurring organic matter, dissolved polymers, fine solid particles. |
| Mechanical Agitation | Physical forces that create and disperse small gas bubbles throughout the liquid, allowing stabilizers to act. | Agitators, pumps, spray nozzles, spargers, turbulent flow in pipes. |
| Liquid Properties | Viscosity, temperature, and composition also influence foam stability. Higher viscosity can trap bubbles more easily, while certain temperatures can promote or inhibit foam. | Viscous glues or paints, temperature fluctuations during production. |
Ultimately, foam is an issue of trapped gas bubbles. Understanding these contributing factors is essential for effective mitigation.
What Are the Harmful Effects of Foam?
Have you experienced the hidden costs and operational nightmares brought on by uncontrolled foam? Its effects go far beyond mere aesthetics, impacting everything from efficiency to safety.
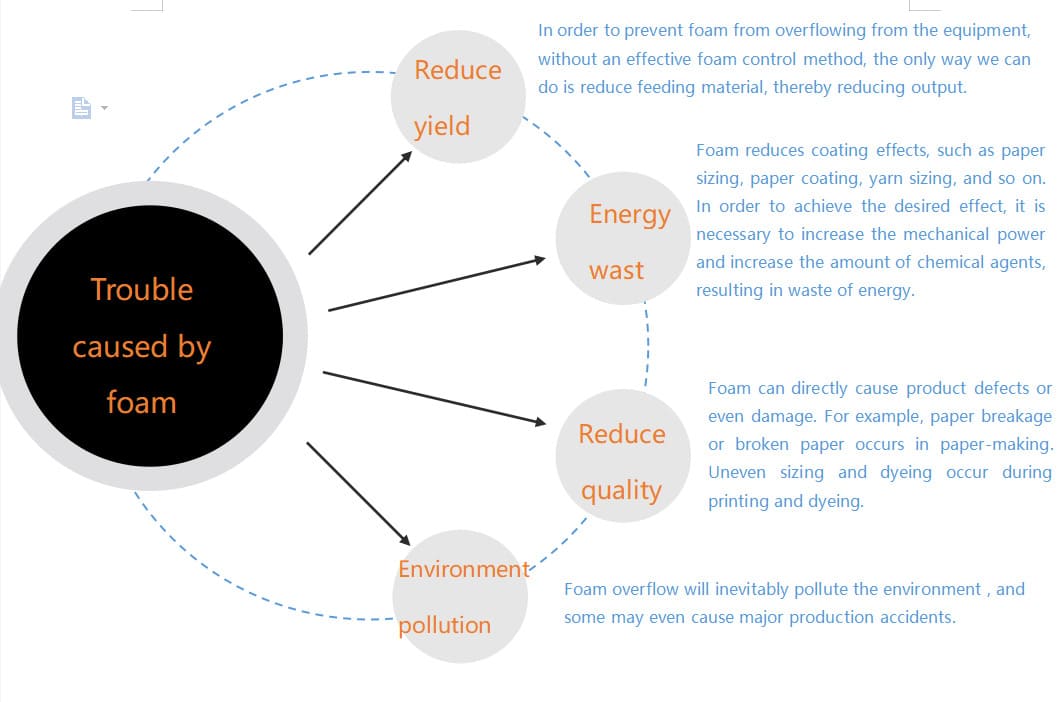
The harmful effects of foam are extensive, causing both direct and indirect problems in industrial processes. Firstly, foam reduces the effective capacity of vessels and tanks. If a reactor or storage tank fills with foam, it means less actual product can be processed or stored, leading to reduced throughput and lost production. For instance, in fermentation, foam can spill over bioreactors, leading to product loss and significant cleaning costs. Secondly, foam can interfere with pumping and filtration. Pumps can become air-locked, causing cavitation and damage, while filter media can become clogged with stable foam, decreasing filtration efficiency and increasing maintenance. Thirdly, it can lead to inaccurate measurements. Foam layers can obscure level sensors, leading to incorrect volume readings and potential overflow or underfilling. Fourthly, it reduces heat transfer efficiency4 in heat exchangers by creating an insulating layer, leading to increased energy consumption5. Finally, foam can create safety hazards6, especially if it contains hazardous chemicals. Operators may be exposed to corrosive or toxic substances during spillages or manual defoaming efforts.
| Aspect Affected | Specific Problem Caused by Foam | Example Industry Consequence |
|---|---|---|
| Capacity & Throughput | Reduces usable volume in tanks, reactors, and vessels. | Fermentation: Decreased batch size, product overflow, costly cleanup, lost yield. |
| Operational Efficiency | Interferes with pumps, filters, and agitation; leads to air pockets and cavitation. | Pumping: Pump damage, reduced flow rates. Filtration: Clogged filters, frequent backwashing. |
| Product Quality | Inaccurate dosing, uneven coating, uneven mixing, entrained air in final product. | Paints/Coatings: Surface defects, pinholes. Agrochemicals: Inaccurate spray application. |
| Safety | Creates spill hazards, slips, and potential exposure to hazardous chemicals. | Chemical processing: Operator exposure, increased cleaning frequency, environmental risks. |
| Maintenance & Downtime | Requires frequent cleaning, potential equipment damage, unscheduled shutdowns. | Wastewater Treatment: Clogged pipes, overflowing clarifiers, pump repair costs. |
| Measurement Accuracy | Obscures level sensors, flow meters, and can lead to incorrect readings. | Food & Beverage: Incorrect filling volumes, off-spec products. |
| Energy Consumption | Reduces heat transfer in heat exchangers, increases mixing energy requirements due to viscous foam. | Textiles: Inefficient dyeing. Cooling Systems: Reduced cooling efficiency. |
In my experience, many clients only see the superficial problem, but the true impact of foam is often much deeper, affecting profitability and sustainability.
How Can We Reduce the Negative Effects of Foam?
Are you looking for practical ways to tackle foam's adverse effects in your operations? Effective foam control involves a combination of strategies, from process adjustments to the right chemical solutions.
To reduce the negative effects of foam, a multi-pronged approach is often most effective, combining process optimization, mechanical solutions, and chemical defoamers7. Firstly, process optimization can involve adjusting parameters such as mixing speed, flow rates, and operating temperatures. Sometimes, simply reducing the agitation intensity or redesigning an inlet pipe to minimize splashing can significantly reduce foam generation. Secondly, mechanical solutions such as physical foam breakers, high-shear mixers, or even vacuum defoaming can be employed. These devices work by physically rupturing foam bubbles, allowing the gas to escape. However, these often require energy and equipment integration. Thirdly, and often most effectively, chemical defoamers or antifoams can be added to the system. Defoamers work by destabilizing the foam lamellae, causing bubbles to coalesce and burst, while antifoams prevent foam from forming in the first place by reducing surface tension and inhibiting bubble stabilization. The choice of defoamer (e.g., silicone-based, oil-based, water-based) depends heavily on the specific liquid system, pH, temperature, and regulatory requirements. It's crucial to identify the root cause of foam and select a solution that addresses that specific mechanism, ensuring it’s compatible with the process and end product.
| Strategy | Description | Application Example |
|---|---|---|
| Process Optimization | Adjusting operational parameters to minimize gas entrainment and foam stabilization. | Reducing agitation speed, optimizing pump selection, redesigning tank inlets to prevent splashing, temperature control. |
| Mechanical Solutions | Utilizing physical devices to break foam bubbles or prevent their formation. | Foam breakers (rotating blades), cyclones, vacuum defoaming systems, redesigned vessel shapes. |
| Chemical Defoamers | Adding chemical agents that either destabilize existing foam (defoamers) or prevent foam formation (antifoams). | Silicone-based defoamers for food processing, mineral oil defoamers for wastewater, non-silicone defoamers for coatings. |
| Raw Material Control | Analyzing and controlling the input materials to ensure they don't introduce excessive foam-promoting agents. | Pre-treating water, sourcing raw materials with lower surfactant content where possible. |
| System Design | Designing equipment and processes with foam prevention in mind from the outset. | Using wider pipes, avoiding sudden drops, ensuring proper venting. |
In my experience, the most impactful foam control strategies are tailored to the specific process, often combining a smart process adjustment with a precisely chosen chemical antifoam.
Conclusion
From my perspective, understanding foam as a process disruptor and not just a nuisance is crucial. By identifying its causes and applying targeted strategies—from process adjustments to advanced defoamer chemistry—we can effectively mitigate its negative impacts, ensuring more reliable, efficient, and profitable industrial operations.
---Discover the connection between chemical reactions and foam generation. ↩
Learn about surfactants and their critical role in stabilizing foam. ↩
Understand how mechanical agitation influences foam stability in liquids. ↩
Discover how foam can hinder heat transfer and increase energy costs. ↩
Learn about the relationship between foam and energy efficiency. ↩
Understand the potential safety risks foam can pose in industrial settings. ↩
Explore effective chemical defoamers that can mitigate foam issues. ↩
